Rechargeable lithium-ion batteries get a lot of headlines, but primary Li battery chemistries are the workhorses in a large number of industrial, medical, consumer, and other applications. This article looks at the performance tradeoffs and typical applications for the six most common Li primary chemistries including LiCFX (lithium poly carbon monofluoride) LiMN02 (lithium manganese dioxide), LiFeS2 (lithium iron disulfate), LiSO2 (lithium sulfur dioxide), LiSOCl2 (lithium thionyl chloride) bobbin and spiral designs, and lithium metal oxide (LMO).
LiCFX
Lithium poly carbon monofluoride, also called lithium/CFx chemistry, batteries can deliver mA currents over long periods of time without a significant voltage drop and are used in applications like pacemakers. The LiCFX chemistry is compatible with the titanium casings that are used for pacemakers. The combination of LiCFX and a titanium case results in about a 50% lower weight compared with the previously used lithium-iodine battery system.
LiCFX also offers high specific energy, low self-discharge, and a wide operating temperature range. LiCFX and hybrid LiCFX-MnO2 cells are suited for use in portable applications, meter reading, and downhole drilling applications. They combine high energy density, continuous low-rate power delivery, and low weight. They are available in prismatic and pouch packages in addition to cylindrical and custom form factors.
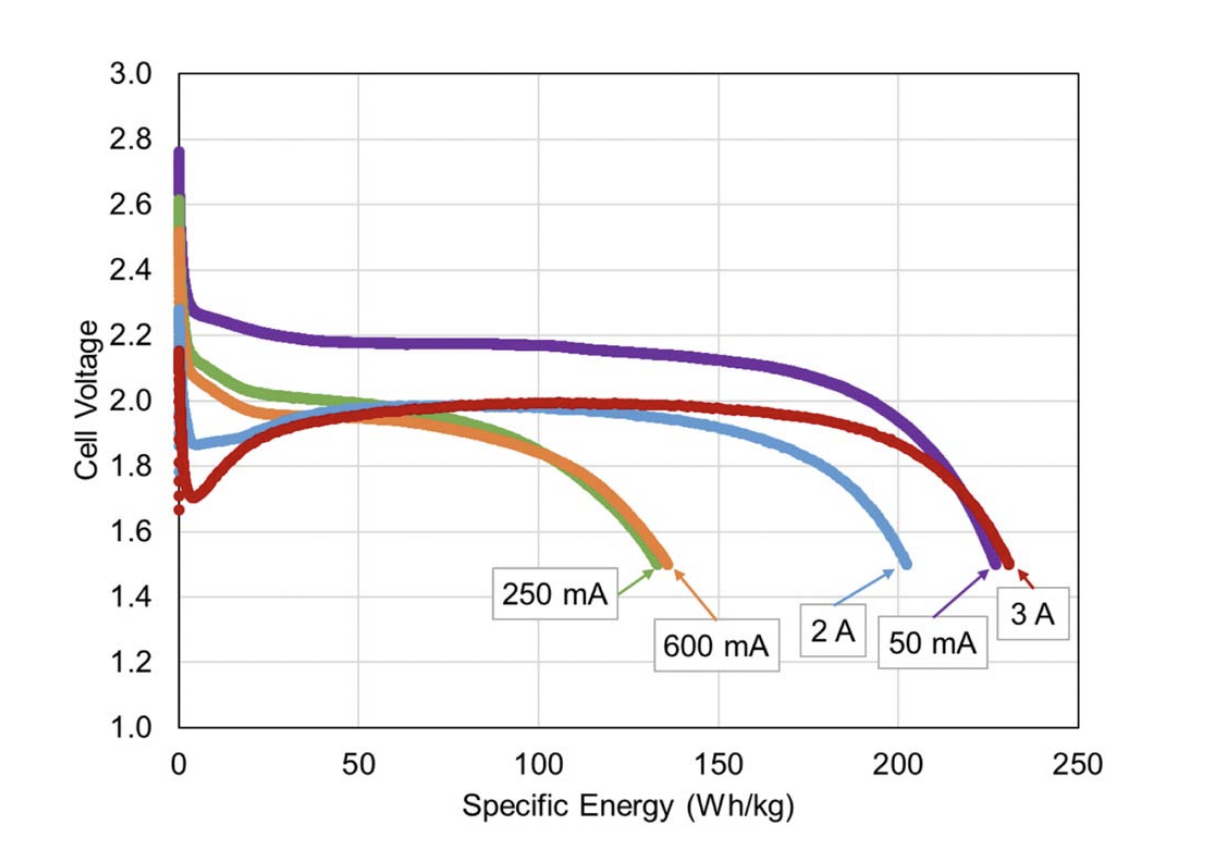
One study of the impact of internal temperature increase in LiCFX hybrid cells found that increasing the discharge rate can result in higher specific energy. For example, the cells in the experiment lost capacity when the discharge current was increased from 50 to 250 ma, but increasing the discharge current to 600 mA or higher resulted in higher available capacity (Figure 1). The test chamber was held at -40 °C. The low discharge currents did not result in an increase in the internal cell temperature. Internal temperatures increased, however, for higher discharge rates, and the higher temperatures are credited with the increase in available energy.
Figure 1. Specific energy vs. discharge rate for Li/CFx-MnO2 cells discharged at −40 °C (Image: Journal of the Electrochemical Society).
LiMN02
Lithium manganese dioxide, sometimes referred to as LiMn, is the most common consumer-grade primary Li battery and accounts for more sales than all other forms of Li primary batteries combined. It’s non-toxic, has good thermal stability, is made with low-cost materials, and is suited for long-life and low-drain applications. It should not be confused with lithium-ion manganese oxide battery (LMO), a rechargeable lithium-ion cell that uses manganese dioxide, MnO2, as the cathode material.
LiMn primary cells provide good energy density. With a nominal voltage of 3 V, these cells deliver about twice the voltage of alkaline or silver oxide batteries. At moderate to high discharge rates, the internal impedance increases, and the terminal voltage declines. LiMn cells are usually rated for operation from -30 to 60 °C. LiMn can deliver high pulse currents but experience corresponding declines in available energy. At temperatures above 25 to 30 °C, self-discharge increases significantly. LiMn cells are available in a wide range of form factors.
LiMn coin cells are often used for applications like real-time clocks and memory backup in embedded systems. The organic electrolyte is highly conductive and supports stable power delivery with little change in operating voltage. LiMn coin cells are available with self-discharge rates of about 1% per year, making them suitable for long-life applications.
LiMn pouch cells can be as thin as 0.4 mm and are designed for use in IoT and wearable devices (Figure 2). Like the coin cells, they have a nominal voltage of 3.0 V. They can support continuous discharge currents up to about 75 mA and pulses up to 150 mA. Pouch cells are rated for operation from -20 to +60 °C.
Figure 2. LiMn pouch cells are designed for use in IoT, wearables, and similar applications (Image: Ultralife).
LiFeS2
Lithium iron disulfate cells are often used to replace primary alkaline cells in consumer applications. They have a longer service life and superior low-temperature operation compared with alkaline, especially in moderate to heavy drain applications. They weigh less than alkaline cells and are environmentally friendly. Compared with conventional alkaline, LiFeS2 cells are high-capacity devices with capacities of 1.1 Ah and 1.8 Ah for AAA and AA cells, respectively.
A typical LiFeS2 cell can be stored at room temperature for up to 15 years and are stable at higher temperatures. The maximum storage and operating temperatures are limited by package considerations. They can be safely stored at 60°C and can tolerate 71°C for at least one week.
Typical LiFeS2 cells include two safety devices. Under very high drain rates, a positive temperature coefficient (PTC) thermal switch limits the current when the temperature reaches 85 to 95 °C. The PTC recloses when the temperature drops to a safe level. In addition, a pressure relief vent opens up at 130 to 160 °C in the event of thermal runaway.
LiSO2
Lithium-sulfur dioxide batteries feature a wide operating temperature range from -55 to +65 °C and are used in challenging environments including defense and marine applications like sonobuoys and life jacket lights, utility metering, mobile asset tracking, security systems, and professional electronics. They have a nominal voltage of 2.9 or 3.0 V (depending on the design), high energy densities, and their spiral construction can deliver repeated high current pulses.
These batteries have a non-flammable (but corrosive) electrolyte. They are pressurized but quickly expel the electrolyte in the case of venting. They feature a self-discharge rate of about 3% per year at 20 °C.
These batteries are available in a variety of sizes including cylindrical formats from 1/2AA to F. A C cell has a capacity of 3.7 Ah, can support a continuous discharge of 100 mA a maximum continuous discharge of 2 A to avoid overheating, and pulses up to 4 A. A D cell has a capacity of 7.8 Ah, can support a continuous discharge of 200 mA a maximum continuous discharge of 2.5 A to avoid overheating and pulses up to 5 A
LiSOCl2
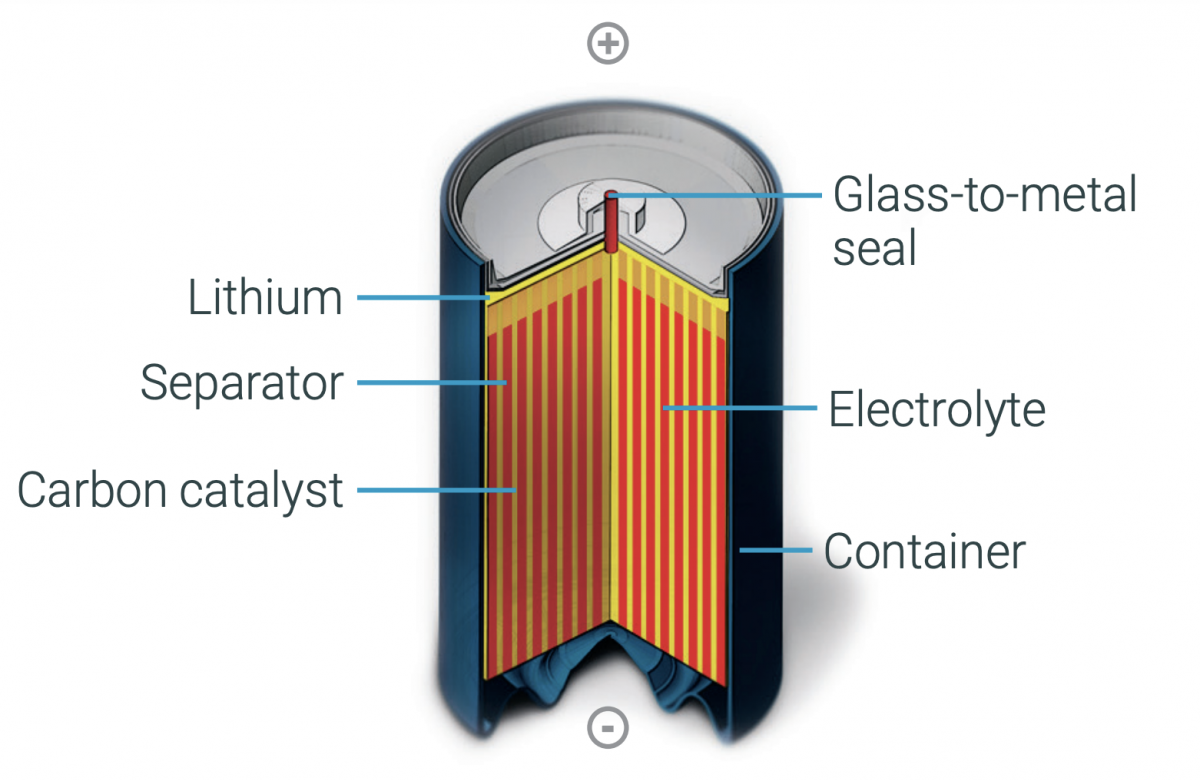
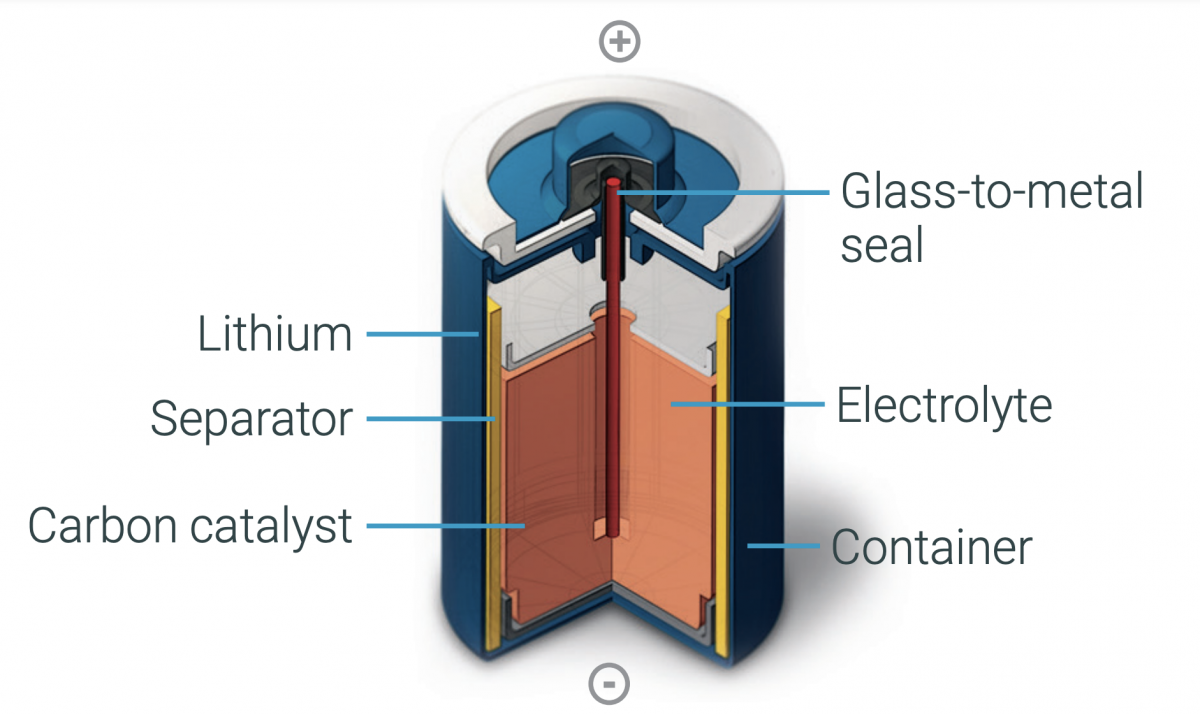
Lithium thionyl chloride batteries are available in the bobbin and spiral wound constructions. Both use a non-aqueous electrolyte that produces a relatively high impedance. Bobbin cells can deliver higher capacities while spiral cells have a larger surface area to deliver higher power densities and can support higher current pulses. Not only does the higher surface area of spiral cells support higher discharge rates, but they also tend to have higher self-discharge rates and shorter operating lifetimes (Figure 3).
Figure 3. LiSOLc2 batteries are available with bobbin (left) and spiral (right) constructions (Image: Saft).
Bobbin-type LiSOCl2 cells are particularly suited for extreme environments and inaccessible locations. They feature a temperature range from -80 to +125 °C, have high energy densities, very low self-discharge, and up to 40-year operating lives. Delivery of high current pulses is not a strength of either spirally wound or bobbin-type LiSOCl2 batteries. A typical high pulse application can demand pulse currents of several amps for an extended period and little or no continuous current flow. Those demands are challenging to support with a standard LiSOCl2 cell, so hybrid solutions have been developed for high-pulse applications.
Hybrids for high pulses
LiSOCl2 hybrid energy storage devices combine a bobbin-type LiSOCl3 cell with a low-impedance hybrid layer capacitor (HCL). Hybrid designs can store up to 700 Wh/kg in the main LiSOCl2 cell and deliver from 280 A/s from small HLCs up to 1,100 A/s using larger HLCs.
These hybrid designs are optimized to support devices with power-hungry two-way communications or remote shut-off functions that need low continuous current levels. Their low self-discharge rate of 1%/year supports operating lives up to 40 years. Since they are based on LiSOCl2 chemistry, these hybrid devices have wide operating temperature capabilities. One challenge can be state of charge (SoC) estimation. As their capacity is depleted, the voltage curve of these batteries plateaus. A circuit can be included to sense the voltage plateau and generate a low battery alert.
LMO
Lithium metal oxide batteries were developed for use in military, medical, and industrial applications that require a lightweight power source that can support high pulses and high rates of continuous power even after extended periods of storage. LMO batteries have been optimized for high-power and medium-power applications and with a self-discharge rate of under 1%/year, can deliver operating lives of up to 20 years.
LMO batteries have a nominal voltage of 4.0 V. High-power LMO batteries are available with capacities from 135 to 500 mAh and can support current pulses from 3.5 to 15 A, respectively. While high-capacity designs deliver 1.4 Ah and can support current pulses up to 9 A. These batteries have an operating temperature range of -55 to 85 °C.
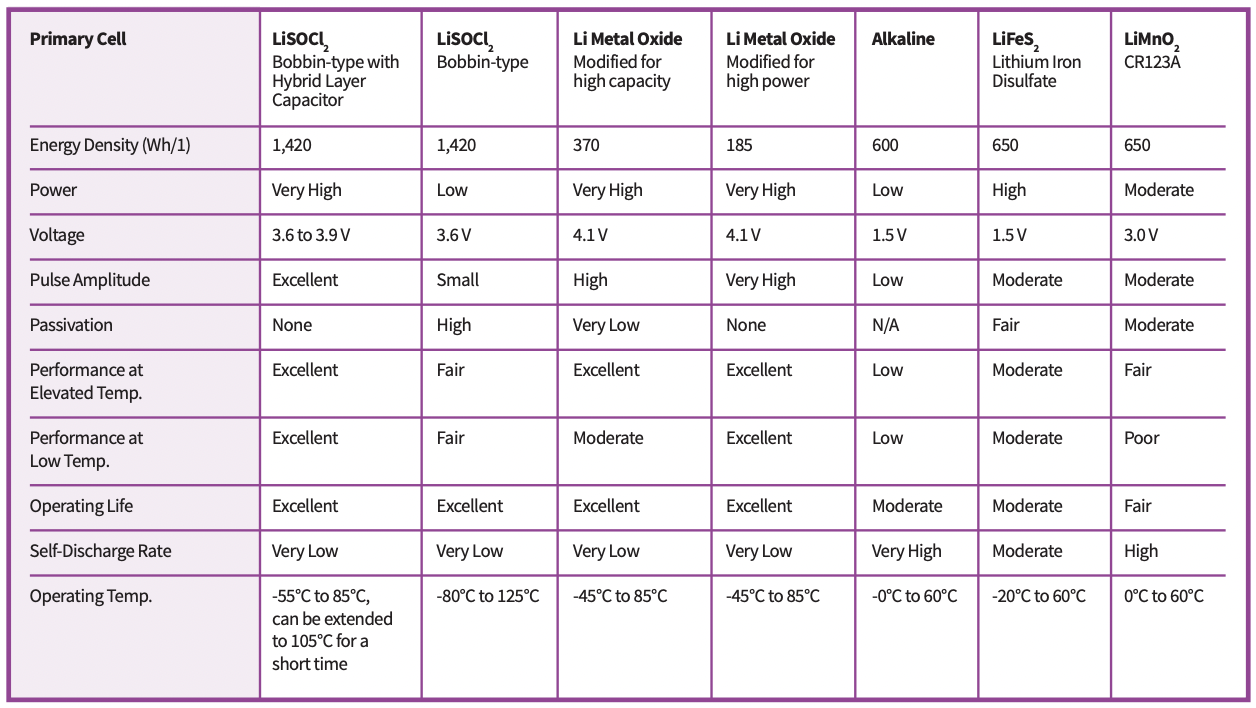
Table 1. Performance comparison of selected primary battery chemistries (Table: Tadiran).
Summary
A wide range of Li primary battery chemistries and constructions are available that have been optimized for applications ranging from cost-sensitive consumer devices to military, medical, and industrial environments where high performance is demanded under harsh conditions. Additionally, primary Li batteries can support 40 years of operation or very high peak pulse power needs.
References
3.0V 700mAh Li-MnO2 non-rechargeable thin cell, Ultralife
Comparing Lithium Chemistries, Tadiran Batteries
How can primary batteries achieve their expected lifetime?, Saft
Lithium Carbon Monofluoride (CFx) and LiCFx-MnO2 Hybrid, Eagle Picher
Lithium Iron Disulfide – Li/FeS2 Application Manual, Energizer
Lithium Manganese Dioxide Battery (Li/MnO2), Maxell
Lithium metal battery, Wikipedia