We interact more and more with glass surfaces than ever before in history. Some people say we have entered the “Glass Age”. Displays, touch screens, touch-enabled glass surfaces, and smart mirrors are finding inroads into our lives. But glass is a tricky material. It can be made tough and flexible, but it sometimes breaks, often with catastrophic effects for the devices we are using. It can be made scratch resistant, but not completely scratch-proof; smudge resistant, but it can still get dirty and even spread infections.
Glass Breaks
However, usually it is not the glass itself that is at fault. It’s the surface defects that are the culprit. They grow and propagate under tension and form cracks. Thin glass – about 100 micrometers thick – without significant surface defects can be rolled up like a plastic film. This shows that glass itself is actually surprisingly strong. The problem is, the glass surface can easily be damaged resulting in the loss of the glass’ inherent strength, unless the defects are prevented from spreading and developing into cracks. If the glass surface is put under sufficient compression such that eventual defects of daily wear and tear will never be under tension, they will not turn into cracks. Thus the inherent strength of the glass is maintained. The glass surface of a Prince Rupert’s drop, for example, is under high compression. The glass drop does not break, even when hit hard with a hammer. You can see this in plenty of demonstration videos on the internet, which also show the impressive (!) result of releasing the compressive stress.
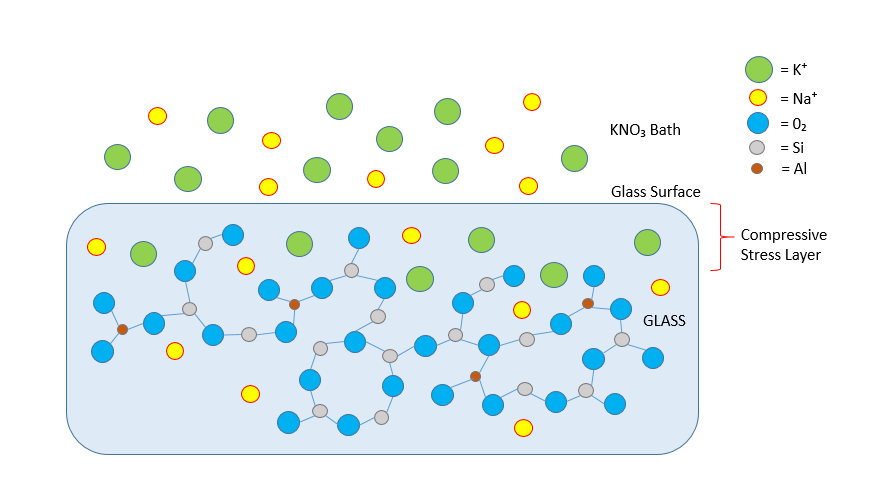
Fortunately, there is a well-known method for creating just the right amount of compressive stress within a glass surface: Chemically speaking, glass is a silicate or in other words a salt of silicic acid. The anion is the silicate ion, which forms a three dimensional structure of silicon and oxygen atoms. The cations can come from the first three groups of the periodic table. If only sodium or potassium are used, the glass dissolves in water. So usually there must be cations of the second or third group like calcium, aluminum, or boron. However, if some sodium or potassium is present, they remain loosely bonded and can be replaced. This is the basis for the chemical strengthening process.Glass which is formulated to have significant sodium in its composition can be strengthened by replacing the sodium ions in the surface layer with larger potassium ions. This is achieved by inserting the glass into a bath of molten potassium salts at greater than 350°C, but below the softening point of the glass. The potassium ions need more space, in the rigid 3-D silicon-oxygen structure, resulting in a highly compressed surface layer of the glass. A compressive stress of well over one giga-pascal can be achieved that way, more than what the remaining center layer of a glass sheet may be able to withstand. Since the surfaces are under compression, there must be an equal but opposite force in the center layer, which is expressed as the tensile stress of the center layer. Too much tensile stress overcomes the molecular structure of the glass, so an ideal amount of surface compression must be obtained in order for hardened glass to perform well. This is dependent on the glass type, the depth and profile of the compressive layer, and the overall thickness of the glass.
Glass Scratches
Glass is hard, but there are everyday items that scratch (dig, chip, cause craters or otherwise damage the surface of) even the hardest glass. An unprotected mobile phone that shared a pocket or purse with keys, coins, pens, or some grains of sand is an unwelcome example. The chemical strengthening process helps to prevent this type of damage by hardening the glass surface so that it is much more scratch resistant than regular glass, although not scratch-proof. Two characteristics of hardened glass are important considerations in the design. First, the depth of the compression layer must exceed the depth of any scratches: As long as a scratch does not penetrate the compression layer, the hardened glass’ strength is maintained. The second important characteristic is the appearance of the scratches. Fine, hardly visible scratches are much less objectionable than wide and clearly visible scratches, occurring when the edges of a scratch chip. A glass formulation with inherent damage resistance that tends towards micro-ductile scratching rather than chipping can be used to avoid the latter.
To avoid scratches, we can protect our expensive devices with screen protectors. Plastic or low cost glass films don’t provide the same benefit as the hardened surface of the original glass lens, but screen protectors made from thin, chemically-hardened glass types are now becoming available and offer much better protection. It’s not possible for these screen protectors to use exactly the same glass material as that of the original hardened lens since a much thinner layer is required, special glass formulations and hardening processes are required to reach similar performance. Furthermore, these screen protectors can be designed such that they break when necessary, thereby absorbing some of the impact energy, and protecting the device to which they are attached. These protectors can easily be replaced after too close of an encounter in a pocket or purse with the aforementioned items or an unfortunate drop onto a gritty sidewalk.
Glass Gets Dirty and Can Spread Infections
When we or others touch our interactive glass surfaces with our fingers or side of our face – as we do when taking calls – we transfer skin oils and micro-organisms onto the touch surface. In addition to the unpleasant look of fingerprints and the degradation of image quality of our high-resolution display this can also lead to spread of infectious diseases. Wouldn’t it be nice to have a glass that is like Teflon, where nothing sticks to it? And wouldn’t it be nice if microbes can’t live on it? This too, is possible with modern glasses.
As mentioned above, the glass structure is a network of silicon and oxygen atoms. But, there are also hydroxyl groups (silicon, oxygen, and hydrogen) on the surface which readily react with certain chemicals like, for example, silyl-chlorides, which are so reactive, that all it takes is to spray the clean surface with a silyl-chloride solution and it chemically binds to the surface. Silyl-chlorides and similar materials can be designed into long molecules with a silyl group on one end and Teflon-like structure on the other. The result is a modified glass surface which rejects oils and fats. When we touch such a surface, less skin oils and microbes get transferred, and whatever may get transferred can easily be wiped off. Such coatings are referred to as “anti-fingerprint” or “oleophobic”. Antimicrobial agents can also be added to the coating or to the glass itself during the chemical hardening process to prevent microbial growth.
The untold truth about thin molecular coatings is that despite their chemical bond to the substrate they wear off eventually, and certainly before the typical end life of an electronic device. This is why it makes more sense to apply these coatings and antimicrobial agents to a hardened glass screen protector which is designed to be replaced several times during the device life.
The right combination of a high quality hardened glass lens with a replaceable hardened glass screen protector that has an oleophobic and antimicrobial coating provides an effective solution for protecting a device from breakage, maintaining the device’s long lasting cosmetic appeal and preventing the spread of infectious diseases via the screen surface. It’s also a versatile solution that can be applied to many applications including smart phones, tablets, ATMs, kiosks, POS terminals and the displays in our cars.

