If you answered 120 over 80 mmHg, you are using criteria that is over 20 years old. As reported in a 2003 article, revised guidelines categorized normal blood pressure as <120/80 mmHg. Since the measurements are meant to lower the risks of not recognizing what is often called the silent killer — high blood pressure — there are other classifications to identify the risk to individuals as well. Prehypertension is 120/80 to 139/89 mmHg, stage 1 hypertension is 140/90 to 159/99 mmHg, and stage 2 hypertension is ≥160/100 or higher mmHg readings. Since “the risk of cardiovascular disease begins at 115/75 mm Hg and doubles with each increment of 20/10 mm Hg,” the 1 mmHg that separates the normal reading from the prehypertension group deserves a closer look.
For those who frequently make pressure measurements in pounds per square inch (psi) or kilopascals (kPa), 1 mmHg is only 0.019 psi or 0.133 kPa. This is a much smaller error value. In blood pressure measurements, the higher value is the systolic blood pressure (SBP) reading and the lower value is the diastolic BP (DBP) reading. Pressure sensors designed for noninvasive blood pressure measurements can have combined linearity and hysteresis error in the 0 to 200 mmHg range of ±0.1% full scale (fs). That is 0.02 mmHg. However, other factors that can cause the error to increase in an application include offset temperature shift and span shift.
In addition to the measurements made in the doctor’s office, people concerned about their blood pressure, especially those diagnosed with prehypertension or more serious conditions, frequently measure their own pressures at home. With many low-cost, automated measurement techniques using microelectromechanical systems (MEMS) and other technologies implemented in a variety of commercial products, one major concern is the validity of the data. To address this problem, the American Medical Association (AMA) created the first U.S. list of blood pressure measurement devices. The US Blood Pressure Validated Device Listing (VDL™), developed to assist physicians and patients in identifying BP devices that have been validated for clinical accuracy, provides greater confidence in the readings and subsequent steps taken by patients and care providers.
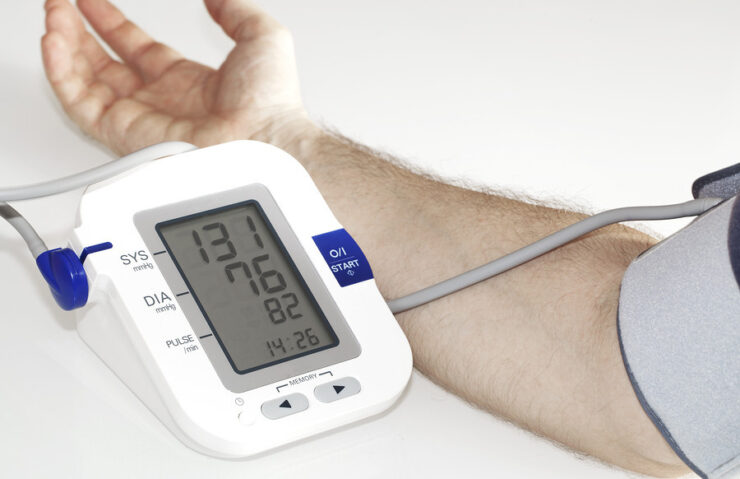 Image source: https://www.health.harvard.edu/heart-health/tips-to-measure-your-blood-pressure-correctly#:~:text=Which%20arm%20should%20you%20measure,is%205%20points%20or%20less
Image source: https://www.health.harvard.edu/heart-health/tips-to-measure-your-blood-pressure-correctly#:~:text=Which%20arm%20should%20you%20measure,is%205%20points%20or%20less
The validation process involves measurement devices cleared by the Food and Drug Administration (FDA) and marketed in the United States under an FDA 510(k) number. Clinical accuracy of a specific make and model is determined based on standards published by the Association for the Advancement of Medical Instrumentation (AAMI), the ANSI/AAMI/ISO standard, and others. Currently, products that make blood pressure readings from the finger and wearable devices using emerging optical or pressure sensor technologies to provide blood pressure readings are not addressed. However, the VDL has 57 clinically accurate devices, including at-home and in-office upper arm devices, wrist cuff devices, community kiosks, and 24-hour ambulatory monitors.
Since a medical professional is typically not involved in the home measurements, the AMA and other organizations provide detailed steps and a process for users to make more accurate and reliable readings. One key step includes choosing an appropriate size (small, standard, large, or extra-large) cuff to make the measurements.
References
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC514035/
Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC7) https://www.nhlbi.nih.gov/files/docs/guidelines/jnc7full.pdf
https://www.med.umich.edu/1libr/UMMG/USBloodPressureVDL.pdf
Blood Pressure Devices, https://www.validatebp.org/